Near infrared-light drug-delivery materials for ocular implants
- OCUMATER -
Project ID: PN-III-P1-1.1-TE-2019-0538
Project leader: Florica Adriana Jerca
Project type: National Funding
Project program: Research projects to stimulate the establishment of young independent research teams – TE competition 2019
Funding Agency: Unit Executive for Funding Higher Education, Research, Development and Innovation
Contractor: "Costin D. Nenitescu" Institute of Organic and Supramolecular Chemistry, Romanian Academy
Start date: 01 September 2020
End date: 31 August 2022
Project abstract:
The present project aims at developing competitive near infrared-light responsive materials for on-demand drug delivery to be used as biodegradable ocular devices. Ocular implants represent up to date solutions developed to treat chronic illnesses of the eye that affect the posterior segment. The most important advantage associated with employing this drug delivery method is the long-term sustained release of drugs at the target location. However, till now there are no ocular implants approved for the on-demand release administration. Poly(2-isopropenyl-2-oxazoline) (PiPOx) is a hydrophilic and biocompatible polymer, which has emerged as a novel and versatile platform to develop advanced functional hydrogel materials, showing high potential to be used in the development of biomaterials. The key aspects that differentiate the protocol of synthesis from other competitive studies is that the polymer crosslinking reaction is clean, does not require a catalyst, and no by-products are formed. From a technological point of view the advantages are associated with the low working temperature, simple technologic process based on water-soluble interchangeable components, and target product specifications. Via the proposed objectives and activities, the multidisciplinary team of young researchers undertook the challenge to demonstrate that PiPOx light-responsive hydrogels may represent a feasible solution in the construction of advanced ocular drug-delivery systems for on-demand release. The team has wide expertise in developing hydrogel materials and light-responsive materials starting from PiPOx, and as well as for in vitro testing.
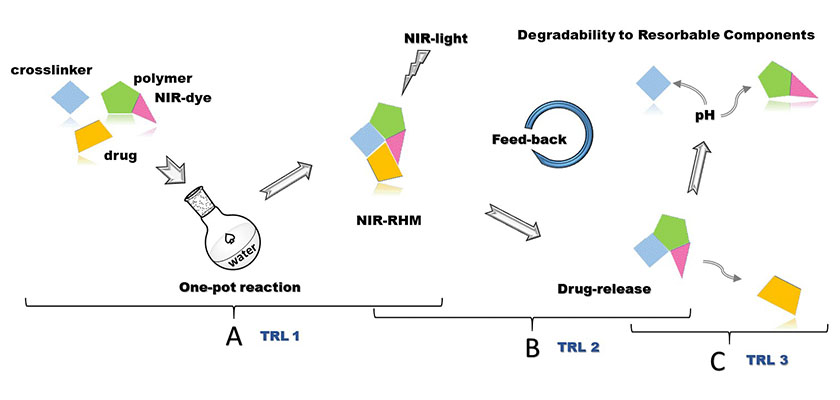
(O1) The first objective aims to optimize the crosslinking protocols of PiPOx with dicarboxylic amino/ hydroxy acids to obtain degradable hydrogel materials with balanced properties. The possibility of mixing all the ingredients in a one-pot strategy towards drug formulations will be tested. This objective corresponds to a TRL 1 (Experimental concept).
(O2) The second objective aims to test and validate the light-assisted protocol for NIR-RHM towards attaining remote control on the drug-release profiles (this objective corresponds to TRL 2, design of Experimental models).
(O3) The third objective aims to validate the drug-release performance of the NIR-RHM in relevant in vitro environments (i.e. simulated body fluids, simulated ocular fluids). From experimental models towards Functional designs to validate the proof of concept for NIR-RHM - this objective corresponds to a TRL 3.
(O4) The fourth objective takes into account the following aspects related to the project: (i) the administrative and scientific risks that meet mitigation solutions in a complete risk management plan; (ii) protection of the intellectual property via a national patent claim; and (iii) dissemination strategies of mid-term outcomes at international level consisting in scientific articles and communications to conferences/congresses.
Project leader:
Dr. Ing. Florica Adriana Jerca
Email: adriana_jerca@yahoo.com, adriana.jerca@ccocdn.ro
Postdoctoral member:
Dr. Ing. Valentin Victor Jerca
Email: victor_jerca@yahoo.com, victor.jerca@ccocdn.ro
Postdoctoral member:
Dr. Ing. Doriana Nicoleta Banu
Email: doriana.banu@yahoo.com, doriana.banu@ccocdn.ro
Postdoctoral member:
Dr. Ing. Diana Elena Giol
Email: diana_giol@yahoo.com
Phd. Student
Emilian Ghibu
Master Student
Ciprian Victor Florea
Summary
The overall goal of the OCUMATER project was to develop competitive near infrared-light responsive hydrogel materials based on the poli(2-izopropenil-2-oxazolina) platform, using an accessible yet versatile approach, to be used for on-demand drug delivery bioresorbable ocular devices. The main goal of the project has been achieved, and the degree of achievement of the specific objectives until August 31, 2022, is 100% according to the funding request of the young teams project, considering the associated risks.
1. The specific objectives of the project:
The first objective aimed to optimize the crosslinking protocols of PiPOx with dicarboxylic amino/ hydroxy acids to obtain degradable hydrogel materials with balanced properties. The possibility of mixing all the ingredients in a one-pot strategy towards drug formulations was tested.
This objective corresponds to a level of technological maturity 1 (Experimental Concept) which derived from the previously published results of our group. To increase the degree of impact on the research results described in the funding request, but also for biological and ecological considerations, the organic solvents previously used in syntheses were replaced with water, and the working temperature was lowered to 60 °C to avoid degradation of the tested drug and the thermal oxidation of amino acids (crosslinkers). Within this objective, the mixing of all ingredients was optimized in one-pot synthesis to obtain drug formulations. The physico-chemical properties, but also the mechanical properties of the resulting hydrogels were optimized. In this context, the determination of the chemical stability of PiPOx under physiological conditions was also carried out, in order to evaluate the applicability of the starting polymer in the biomedical context, which resulted in a first ISI scientific paper published in a specialized journal from ACS. Another scientific paper will be submitted for publication based on the experimental concept.
The second objective aimed to test and validate the light-assisted protocol for NIR-RHM towards attaining remote control on the drug-release profiles.
This objective corresponds to a technological maturity level 2 (Experimental Models). Based on the previous experience in the characterization of light-sensitive functional materials, the response to the light stimulus of the developed hydrogels was optimized, obtaining the release of the drug on demand but also reversibility within the investigated systems. In this context, a review paper was elaborated on the topic of the project to emphasize the relevance of using azobenzene derivatives sensitive to red light in the construction of biodegradable materials that release drugs for emerging fields such as photopharmacology. A second scientific paper was published in a specialized journal from Nature with high FI.
The third objective aims to validate the drug-release performance of the NIR-RHM in relevant in vitro environments (i.e. simulated body fluids, simulated ocular fluids).
This objective corresponds to a technological maturity level 3 (Functional Models). Based on the previous experience in investigating stability and degradability properties, hydrogels with slow or accelerated degradable profile were obtained depending on the nature of the crosslinker used. This aspect is important because it dictates the resorbable character of the implant over time. Within this objective, the interaction of the raw materials, the intermediates, and the final hydrogels with different cell lines was determined, which contributed to a differentiation of the level of cytotoxicity in all experimental stages. In this context, the experimental lab technology to produce PHOTOSENSITIVE AND DEGRADABLE POLYMERIC HYDROGELS BASED ON POLY(2-ISOPROPENYL-2-OXAZOLINE) AND THERAPEUTIC AGENTS was realized. To protect intellectual property rights, a national patent application was submitted to OSIM.
The fourth objective consisted in disseminating the results of the project and protecting intellectual property, integrated into a managerial plan focused on scientific risk management.
Within this final objective, the following aspects related to the project were considered: (i) administrative and scientific risks that were accompanied by mitigation solutions in a complete management plan; (ii) intellectual property protection through a national patent claim; and (iii) by dissemination of the preliminary scientific results at the international level consisting of 3 ISI rated scientific papers with IF > 4, and 4 scientific communications at international conferences.
2. Activities:
Stage I/2020
Activity 1.1. Protocols for the preparation of reference materials and physico-chemical characterization methods used in obtaining hydrogels have been established. (2020)
Stage II/2021
Activity 2.1. Protocols for the preparation of reference hydrogels and physico-chemical characterization methods have been optimized. (2021)
Activity 2.2. Protocols for the preparation of photosensitive hydrogels and physico-chemical characterization methods have been established. (2021)
Activity 2.3. The response time to the near infrared light of the reference materials was investigated. (2021)
Activity 2.4. The near-infrared light response time of photosensitive hydrogels was investigated. (2021)
Activity 2.5. The protocols for obtaining the reference hydrogels and the photosensitive hydrogels have been perfected. (2021)
Activity 2.6. Dissemination through international communication and publication of preliminary results has been successfully accomplished. (2021)
Stage III/2022
Activity 3.1. The degradability of reference and photosensitive hydrogels was investigated.
Activity 3.2. The drug-release of hydrogels in the presence and absence of light stimulus was investigated and optimized. (2022)
Activity 3.3. Cytotoxicity evaluation of the reference and photosensitive hydrogels was investigated. (2022)
Activity 3.4. Dissemination of the results by international communication and publishing has been successfully accomplished. (2022)
Publications
1. Jerca, F.A.; Jerca, V.V.; Hoogenboom, R.; “In vitro assessment of the hydrolytic stability of poly(2-isopropenyl-2-oxazoline)”, Biomacromolecules 2021, 22 (12): 5020–5032, impact factor 6.988.
https://doi.org/10.1021/acs.biomac.1c00994
2. Jerca, F.A.; Jerca, V.V.; Hoogenboom, R.; “Advances and opportunities in the exciting world of azobenzenes”, Nature Reviews Chemistry, 2022, 6: 51–69, impact factor 34.035.
https://doi.org/10.1038/s41570-021-00334-w
3. E. Ghibu, E.D. Giol, I. Caraș, V. Vasile, N.D. Banu, D.M. Vuluga, R. Stan, V.V. Jerca, F.A. Jerca, “Poly(2-isopropenyl-2-oxazoline) hydrogels with tunable degradation profiles for drug delivery applications”, to be submitted.
Patent claim:
F.A. Jerca, V.V. Jerca, E. Ghibu, N.D. Banu, HIDROGELURI POLIMERICE FOTOSENSIBILE ȘI DEGRADABILE PE BAZĂ DE POLI(2-IZOPROPENIL-2-OXAZOLINĂ) ȘI AGENȚI TERAPEUTICI ȘI PROCEDEU DE OBȚINERE AL ACESTORA, no. A2022/ 00386 with deposit date 05.07.2022.
Conferences
1. E. Ghibu, N.D. Banu, D.M. Vuluga, R. Stan, F.A. Jerca, V.V. Jerca, “Degradable hydrogels for drug delivery applications based on poly(2-isopropenyl-2-oxazoline)”, 2nd Bucharest Polymer Conference, București, România, 10 –11 iunie 2021. Prezentare orală
2. E. Ghibu, C.V. Florea, N.D. Banu, D.M. Vuluga, V.V. Jerca, F.A. Jerca, “Poly(2-isopropenyl-2-oxazoline) hydrogels with tunable degradation profiles”, 15th International Conference on Materials Chemistry (MC15), 12 –15 iulie 2021. Poster ONLINE
3. E. Ghibu, N.D. Banu, D. Giol, V.V. Jerca, F.A. Jerca, “Degradable hydrogels for drug delivery applications based on poly(2-isopropenyl-2-oxazoline)”, 32nd Edition of the international congress “PREPARING THE FUTURE BY PROMOTING EXCELLENCE”, 28 feb-2 mar 2022 Iasi. Prezentare orală ONLINE
4. E. Ghibu, N.D. Banu, C.V. Florea, V.V. Jerca, F.A. Jerca, “Degradable hydrogels for drug delivery applications based on poly(2-isopropenyl-2-oxazoline)”, The 49th World Polymer Congress, Winnipeg, Canada, 17- 21 Iulie. Poster ONLINE
Awards:
Emilian Ghibu: Best oral presentation award for “Degradable hydrogels for drug delivery applications based on poly(2-isopropenyl-2-oxazoline)” acordat în cadrul conferinței 2nd Bucharest Polymer Conference, UPB, sponsorizat de POLYMERS a Multidisciplinary Digital Publishing Institute (MDPI) of open access scientific journals, 300 €.





