- Project Home
- Objectives
- Project progress
- Dissemination
- Members
- Cognitiv and socio-economic impact
- Contact
Redox-active azulene-based organic cages and metalla-cages in crystal engineering
(AZMETCA)
Contract Authority: Executive Agency for Higher Education, Research, Development and Innovation Funding (UEFISDCI)
Funding programme: PNCDI IV - Program 5.8 - Cooperare Europeană și Internațională
Subprogram: Bilateral/Multilateral - Proiecte de Colaborare cu Republica Moldova
Project code: PN-IV-P8-8.3-ROMD-2023-0045
Acronym: AZMETCA
Time span: 05/2023 – 05/2026
Project abstract: Development of porous self-assembled materials, like of porous organic cages (POCs) able to recognize and host molecules, by judicious selection of the constituting building blocks is highly desirable in modern material science. Depending on the properties of the selected building blocks, the target compound can be conveniently endowed with various cooperative properties.
In this regard, electroactive and/or photochromic entities are promising candidates to access multifunctionality, since upon external stimuli a direct control of their structures and, thus of their resulting properties is possible. Their relevance is more increased when the attached cromophores bring additional properties. We have shown that the attachment of azulene moieties on this photochromic unit allows the photocyclization by irradiation with 405 nm visible light (https://doi.org/10.1039/C5RA11974J). These compounds function as fluorescent switches, exhibiting dramatic decrease in the azulene S2→S0 fluorescence emission after photocyclization. Furthermore, the cyclization reaction is also induced by electrochemical oxidation.
Our goal is to design multifunctional photochromic materials, by incorporating the azulene derivatives with substituents rationally selected into discrete organic- and metal-organic cages. The output of the project is represented by the rational design of discrete organic- and metal-organic cages able to bind a specific guest, and, subsequently, to release the guest molecule through an external stimuli.
Objectives
The project seeks to combine the principles of photocomutation processes with crystal engineering approaches, to develop compounds of interest in material science, focusing in particular on fluorescent, redox and porous properties. Two families of molecular cages will be obtained using original, electroactive and photochromic azulene derivatives as tectons.
The objectives are the following:
- Development of electroactive organic and metal-organic cages via Schiff base condensation reaction between tris-amines with different flexibility of methyleneaminic chains and azulene-dialdehydes.
- Self-assembly of photoswitchable organic and metalla- cages, employing azulenyl-dithienylcyclopentenes (perhydro and perfluoro) as carbonyl reactants for [2+3] and [4+6] Schiff condensations reaction.
The project is focused onto bringing together the strong expertise of the research groups in organic synthesis of azulene derivatives and organic/metal-organic assemblies (Romania), and computational chemistry (Moldova), resulting into a fruitful exchange of expertise.
Project progress
New organic cages were built from the reaction of tris(2-aminoethyl)amine and azulene-1,3-dicarboxaldehyde through Schiff base condensation reaction. The equimolecular reaction of the dialdehyde with the tris-amine, in the presence of trifluoroacetic acid, yielded the cationic [1+1]2+ tetraimine cage, 1, whereas a 3 : 2 ratio lead to the cyclic hexaimine cage, 2, both in presence or in the absence of the trifluoroacetic acid.
The molecular structure determinations show π-π interactions established between azulene moieties, intra-cage in the tetraimine structure and inter-cage in the cyclic hexaimine compound. The cryptand type hexaimine cyclic cage can accommodate silver cations through a template synthetic approach, resulting a disilver-metallacage. The complexation induces significant changes in the conformation of the macrobicycle, from eclipsed to a staggered orientation of the bridgehead N-CH2- bonds. Furthermore, the metallacage assembles into a supramolecular network with a honeycomb topology via π-π stacking interaction between azulene moieties. The hexaimine cage functions as halogen bond acceptor forming halogen-bonded chains with triiodoperfluorobenzene. Owing to the azulene motif, these cages show fluorescent properties, especially in solid state, the intensity and position of the emission peaks depending on the molecular environment and solid-state packing of the discrete molecules.
The synthesis of photoswitchable organic cages constitutes an emerging field within supramolecular chemistry, in which molecular systems are engineered to assemble into closed three-dimensional architectures capable of reversibly modulating their shape, polarity, or internal cavity accessibility upon light irradiation. To achieve this objective, novel dithienylcyclopentene derivatives functionalized with azulenyl moieties bearing electron-withdrawing substituents were synthesized. Their photochromic, fluorescent, and electrochemical properties were systematically investigated. The results revealed that the fluorescence intensity is strongly influenced by the nature of the substituent on the azulene unit, and that photocyclization of the dithienylcyclopentene core leads to a significant enhancement of fluorescence as a result of extended π-conjugation.
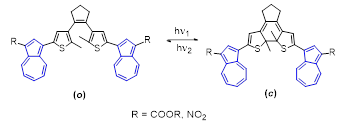
The photoswitching behavior was monitored by UV/Vis spectroscopy, and the results are illustrated in the figure below for R = COOEt and R = NO₂. In dichloromethane, the initial absorption spectra display two intense bands in the UV region at approximately 230 and 290 nm, together with a broad shoulder between 300–330 nm, as well as a band in the visible region at 405–442 nm, which is significantly red-shifted in the case of the nitro-substituted derivative. Upon irradiation, for the compound substituted with –COOEt, the intensity of the absorption maximum at 317 nm decreases progressively, accompanied by the formation of a new band at approximately 306 nm. An isosbestic point at 344 nm can be observed, indicating an equilibrium between the two species: the open and closed forms.
In the case of the nitro-substituted system, photoswitching upon irradiation is slower. In comparison with the previous examples, two isosbestic points can be observed in this case, indicating a more complex equilibrium in solution.
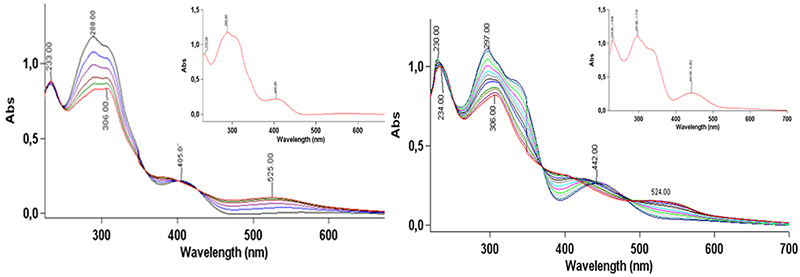
UV–vis spectrum of the compound in its initial state (inset) and monitored upon irradiation for 150 seconds for R = COOEt (left) and 360 seconds for R = NO₂ (right).
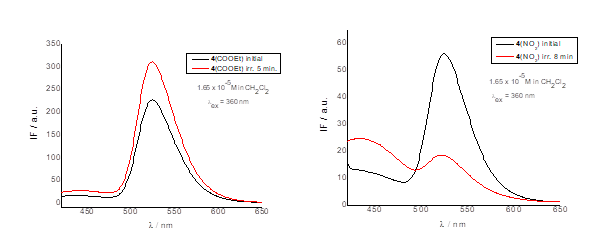
The compounds are fluorescent, exhibiting emission at 524 nm upon excitation at a wavelength of 360 nm. It was observed that the fluorescence intensity depends on the substituent attached to the azulene moiety, with strong quenching in the case of the R = NO₂ compound.
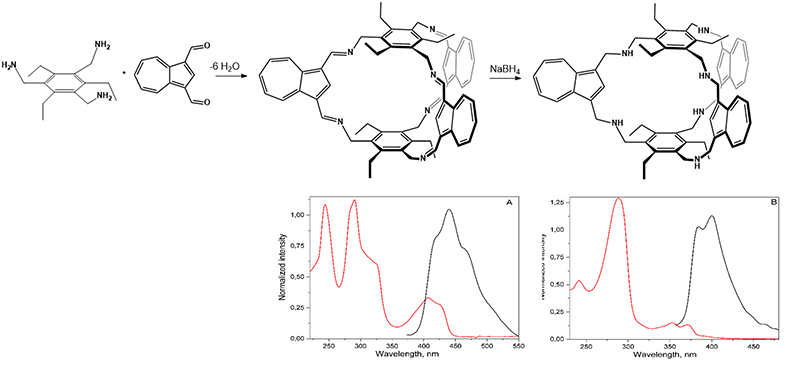
 Using azulene dialdehyde as the linker and 1,3,5-tris(aminomethyl)-2,4,6-triethylbenzene as the triamine node, two types of macrobicyclic cages were isolated. A new hexaimine [2+3] cage was obtained via Schiff-base condensation, exhibiting a symmetric structure both in solution and in the solid state. X-ray diffraction analysis revealed a two-dimensional architecture assembled through π–π and C–H···π interactions involving the azulene units.The hexaimine organic cage crystallizes as green rhombic crystals in the space group P2₁/n. The two phenyl groups of the 1,3,5-tris(aminomethyl)-2,4,6-triethylbenzene moiety define a central cavity of approximately 9.8 Å. The imine groups are oriented on the same side of the benzene ring, while the ethyl groups point outward, toward the exterior of the cavity. Inside the molecular cage, the bis-imine units adopt an anti,anti conformation, and the cage exhibits an eclipsed, helical-type geometry.
Using azulene dialdehyde as the linker and 1,3,5-tris(aminomethyl)-2,4,6-triethylbenzene as the triamine node, two types of macrobicyclic cages were isolated. A new hexaimine [2+3] cage was obtained via Schiff-base condensation, exhibiting a symmetric structure both in solution and in the solid state. X-ray diffraction analysis revealed a two-dimensional architecture assembled through π–π and C–H···π interactions involving the azulene units.The hexaimine organic cage crystallizes as green rhombic crystals in the space group P2₁/n. The two phenyl groups of the 1,3,5-tris(aminomethyl)-2,4,6-triethylbenzene moiety define a central cavity of approximately 9.8 Å. The imine groups are oriented on the same side of the benzene ring, while the ethyl groups point outward, toward the exterior of the cavity. Inside the molecular cage, the bis-imine units adopt an anti,anti conformation, and the cage exhibits an eclipsed, helical-type geometry.
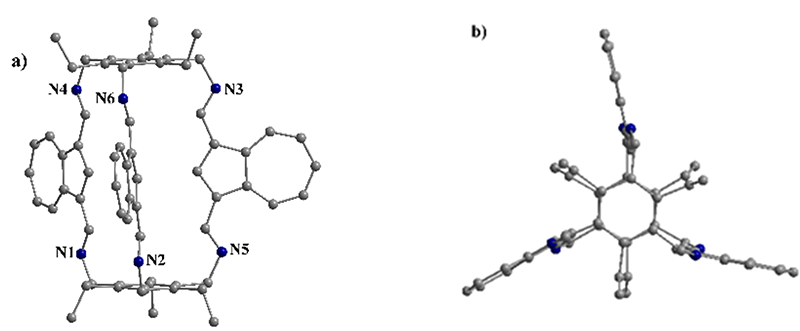
Due to the presence of azulene in the molecule, the molecular organic cage absorbs in the visible region and exhibits fluorescence. Reduction with NaBH₄ led to the isolation of the corresponding macrobicyclic cage with a hexamine structure. The reduced cage displayed a hypsochromic shift and a decrease in fluorescence intensity, indicating that the reduction of the imine groups to amines disrupts the conjugated system, resulting in shorter absorption and emission wavelengths. Both cages are redox-active, with the hexamine structure exhibiting higher current values, likely due to improved electron-transfer kinetics facilitated by its more flexible chemical structure.
Team members
Coordinator: "C. D. Nenitzescu" Institute of Organic and Supramolecular Chemistry, Bucharest, Romania
Team:
Prof. Marius Andruh, project director
Dr. Simona Nica, researcher
Dr. Anamaria Hanganu, researcher
Dr. Adrian E. Ion, researcher
Dr. Andreea Horga, postdoctoral researcher
Partner: State University, Institute of Chemistry, Chisinau, Moldova
Team:
Acad. Gheorghe Duca, partner leader
Dr. Natalia Bolocan, postdoctoral researcher
Dr. Vladislav Blonschi, postdoctoral researcher
Dr. Iolanta Balan
Crina Vicol, Ph. D. student
Carolina Grigoras, Ph.D. student
EN
Cognitive Impact:
The project advances fundamental knowledge on the assembly of complex structures with interesting photophysical and electrochemical properties. High-yield synthetic protocols have been developed, which can be extended to other similar chemical systems. The results have been disseminated through four scientific articles and presentations at international conferences, strengthening the scientific achievements of the involved teams, both at national and international levels.
Socio-Economic Impact:
The project supports the training of early-career researchers, through the direct involvement of a PhD student and a postdoctoral researcher from the partner team of the Moldova Republic. Through research stays, team members acquire skills in experimental methodologies and critical data analysis. In the long term, this contributes to the development of human capital in the field of chemical sciences and advanced materials, creating opportunities for future collaborations in academic and/or applied research. Furthermore, the project facilitates knowledge exchange , and strengthens academic and scientific collaboration between the two countries.
_
RO
Impact cognitiv: avansarea cunoașterii fundamentale privind asamblarea structurilor complexe cu proprietăți fotofizice și electrochimice interesante. Au fost dezvoltate protocoale de sinteza cu randamente mari care pot fi extinse si catre alte sisteme chimice similare. Rezultatele obținute au fost diseminate in cadrul a patru articole stiintifice si prezentări în cadrul conferințelor internaționale, consolidând poziția științifică a echipei la nivel național și internațional.
Impact socio-economic: Proiectul susține formarea tinerilor cercetători prin implicarea directă a unei doctorande și a unei postdoctorande din echipa parteneră din Republica Moldova. Prin stagiile de cercetare realizate, membrii echipei dobândesc competențe în metodologii experimentale și analiză critică a datelor. Pe termen lung, acest proces contribuie la dezvoltarea capitalului uman în domeniul științelor chimice și materialelor avansate, creând premise pentru viitoare colaborări în mediul academic și/sau aplicativ. În plus, proiectul conferă schimbul de cunoștințe intre echipele implicate, precum și consolidarea colaborarii intre cele doua tari.
Contact
"C. D. Nenizescu" Institute of Organic and Supramolecular Chemistry
Splaiul Independentei 202B, Bucharest, Romania, 060023
Prof. Marius Andruh
email: marius.andruh@acad.ro







